Exploring the Ionization Energy Trend: A Simple Guide to Atoms
Have you ever wondered why some things stick together while others pull apart easily? In the world of science, atoms have a similar struggle. Ionization energy is the specific amount of “muscle” or energy needed to pull an electron away from an atom. Think of it like a game of tug-of-war. Some atoms hold onto their electrons very tightly, while others let them go without much of a fight. When we talk about the ionization energy trend, we are looking at the patterns of this energy across the periodic table. Understanding this concept helps scientists predict how different elements will react when they meet each other in a lab or in nature.
I remember first learning about this in school and feeling a bit confused. It helps to imagine atoms as little magnets. The closer the “magnet” pieces are, the harder they are to pull apart. This simple idea is the secret to mastering the ionization energy trend. By looking at where an atom sits on the chart, you can guess if it is a “hoarder” of electrons or a “giver.” This makes chemistry feel much more like a puzzle and less like a chore.
Table of Contents
How the Ionization Energy Trend Works Across Rows
When you look at the periodic table, you see rows called periods. As you move from the left side to the right side, the ionization energy trend usually goes up. This happens because atoms get more protons in their center as you move right. Protons are like tiny anchors that pull on the electrons. With more protons, the pull gets stronger, making it much harder to steal an electron away. This is why elements on the far right, like Neon, are very stable and don’t like to share.
On the left side, elements like Lithium have fewer protons pulling on their outer electrons. Because the pull is weaker, the ionization energy trend stays low there. These atoms are often very reactive because they lose electrons so easily. If you are trying to remember this for a test, just think: “Moving right makes it tight!” The tighter the hold, the higher the energy you need to break it. It is a very consistent pattern that helps us organize all the elements we know today.
Why Does Energy Change as You Move Down?
Now, let’s look at what happens when we move down a column, also known as a group. In this case, the ionization energy trend actually goes down. You might think that more protons would make the pull stronger, but there is a catch. As you go down the list, atoms get “fatter” because they add more layers of electrons. These extra layers act like a shield. The outer electrons end up being very far away from the center, so the pull isn’t as strong as it used to be.
I like to compare this to a big stadium. If you are sitting in the very front row near the stage (the nucleus), you can hear the music perfectly. But if you are in the very last row of a massive stadium, the sound is much weaker. In the ionization energy trend, those “back row” electrons are easy to snatch away because they barely feel the pull from the center. This explains why elements at the bottom of the table are often the most eager to react and change.
The Role of the Nucleus in Atomic Energy
The nucleus is the heart of the atom. It is filled with protons that have a positive charge. Since electrons have a negative charge, they are naturally attracted to the nucleus. This attraction is the main engine behind the ionization energy trend. When the nucleus is strong and close to the electrons, the energy required to remove one is very high. Scientists call this “effective nuclear charge.” It is a fancy way of saying how much power the center actually has over its orbiting friends.
When we study the ionization energy trend, we are really studying the strength of this central “magnet.” Elements with a very high nuclear charge relative to their size will always have a higher ionization energy. It is like trying to take a toy away from a very strong toddler versus a sleepy one. The stronger the “toddler” (the nucleus), the more effort you have to put in. This fundamental rule makes the periodic table a very predictable and useful tool for everyone from students to professional chemists.
Key Facts About Chemical Elements
| Element Name | Atomic Symbol | Trend Position | Energy Level |
| Helium | He | Top Right | Very High |
| Francium | Fr | Bottom Left | Very Low |
| Fluorine | F | Top Right | High |
| Cesium | Cs | Bottom Left | Low |
| Oxygen | O | Right Side | High |
| Sodium | Na | Left Side | Low |
Understanding Electron Shielding Simply
We mentioned “shielding” earlier, and it is a big part of the ionization energy trend. Imagine you are wearing five heavy coats. It would be hard for someone to feel your heartbeat through all that fabric, right? In an atom, the inner electrons act like those coats. They block the “heartbeat” (the pull) of the nucleus from reaching the outer electrons. This is why the ionization energy trend drops as atoms get larger and add more shells.
This shielding effect is the reason why some big atoms are so “lazy.” They have plenty of protons, but those protons are hidden behind so many layers that they can’t hold onto their outer electrons. When you see the ionization energy trend dip, you are usually seeing the result of shielding in action. It is one of the most important concepts to grasp if you want to understand why the periodic table is shaped the way it is.
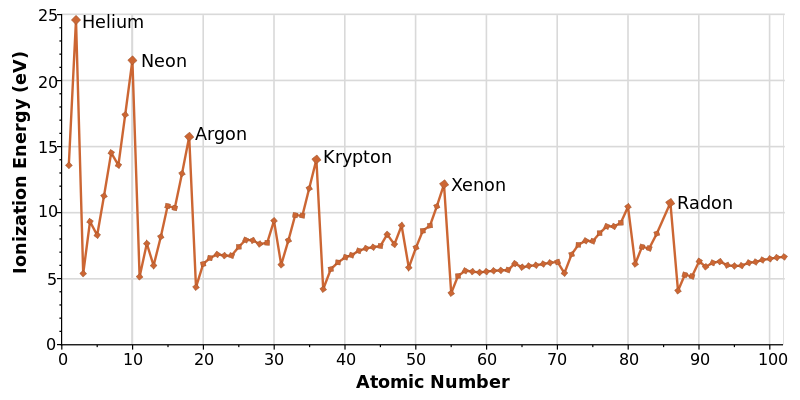
Why Helium Has the Highest Ionization Energy
Helium is the king of the ionization energy trend. It sits at the very top right of the table. It is a tiny atom with two protons and two electrons in a single shell. Because the electrons are so close to the nucleus and there is zero shielding, Helium holds onto them with incredible strength. You would need a massive amount of energy to pull an electron away from a Helium atom. This is why Helium is an “inert” gas—it just doesn’t want to react with anyone.
Looking at Helium helps us see the peak of the ionization energy trend. It represents the perfect storm of a strong nucleus and a very short distance. In my own studies, I found that using Helium as a benchmark made everything else easier to understand. If you know why Helium is at the top, you can easily figure out why elements on the opposite side, like Francium, are at the bottom. It provides a clear “high point” for the entire map of elements.
Exceptions to the General Energy Rules
Sometimes, the ionization energy trend has a few “hiccups.” Nature isn’t always perfect! For example, you might see a small drop in energy between certain groups. This usually happens because atoms like to have their electron shells half-full or completely full. It is like a person feeling more balanced when they have both hands full instead of just one. When an atom is in a stable “balanced” state, it might resist the general ionization energy trend just a little bit.
These exceptions are what make chemistry so interesting. While the ionization energy trend gives us a great general map, these tiny details show us the deeper personality of each element. If you see a spot where the energy doesn’t go up as expected, don’t worry! It’s just the atom trying to stay comfortable and stable. Learning these small quirks will actually help you become an expert much faster than just memorizing the main rules.
Why This Trend Matters in Real Life
You might be thinking, “Why should I care about the ionization energy trend?” Well, it actually affects the world around you! For example, the batteries in your phone often use Lithium. We use Lithium because it has a low ionization energy trend value, meaning it gives up electrons very easily. This flow of electrons is exactly what creates electricity! Without understanding these energy patterns, we wouldn’t have the portable tech we use every single day.
From the salt on your dinner table to the metal in your car, the ionization energy trend determines how elements bond together. It tells us which metals will rust quickly and which ones will stay shiny. By mastering this concept, you are basically learning the “social rules” of the universe. It helps us build better materials, cleaner energy, and safer medicines. Science is all about patterns, and this is one of the most useful patterns we have ever discovered.
How to Predict Atomic Behavior Easily
Once you know the ionization energy trend, you can start predicting how atoms will behave without even looking it up. If someone tells you an element is in the bottom-left corner, you immediately know it has low energy and loves to lose electrons. If they point to the top-right, you know it’s a “tough” atom that holds on tight. This mental map is the ultimate shortcut for any science student or curious mind.
I always tell people that the ionization energy trend is like a weather map for atoms. Just like a map tells you where it might rain, the periodic table tells you where the “energy storms” are. It simplifies a very complex subject into a few easy directions: up and to the right for more energy, down and to the left for less. With this knowledge, you can walk into any chemistry conversation with confidence and a clear understanding of how the world works at a tiny level.
Frequently Asked Questions
1. Does ionization energy increase or decrease across a period?
The ionization energy trend increases as you move from left to right across a period. This is because the nucleus gets more protons and pulls harder on the electrons.
2. Which element has the lowest ionization energy?
Francium is generally considered to have the lowest value in the ionization energy trend. It is a very large atom, so its outer electrons are very far from the center.
3. Why do noble gases have such high energy?
Noble gases like Neon and Helium have a full outer shell. They are very stable and “happy,” so they follow the ionization energy trend by resisting any attempt to remove their electrons.
4. What is the difference between first and second ionization energy?
The first ionization energy trend refers to removing the first electron. The second refers to removing a second one. Usually, it takes much more energy to remove a second electron!
5. How does atomic radius affect this trend?
As the atomic radius gets smaller, the ionization energy trend goes up. This is because the electrons are closer to the “magnetic” pull of the nucleus.
6. Can temperature change the ionization energy?
While the base ionization energy trend is a property of the atom itself, adding heat can give electrons more energy, making them easier to move in a lab setting.
Conclusion: Mastering the Energy Map
Understanding the ionization energy trend is like finding the “cheat code” for the periodic table. It takes a wall of confusing letters and numbers and turns it into a logical, flowing story. We have learned that atoms pull on electrons based on how many protons they have and how many “shields” are in the way. By remembering that energy increases as you go up and to the right, you can predict the behavior of almost any element on the chart.
Whether you are studying for a big test or just curious about how your phone battery works, this trend is a vital piece of the puzzle. Science doesn’t have to be hard when you look for the patterns! I hope this guide made the ionization energy trend feel simple and even a little bit fun. Keep exploring, stay curious, and remember that even the smallest atoms follow these amazing rules. Now, why not take a look at a periodic table and see if you can spot the “energy kings” yourself?